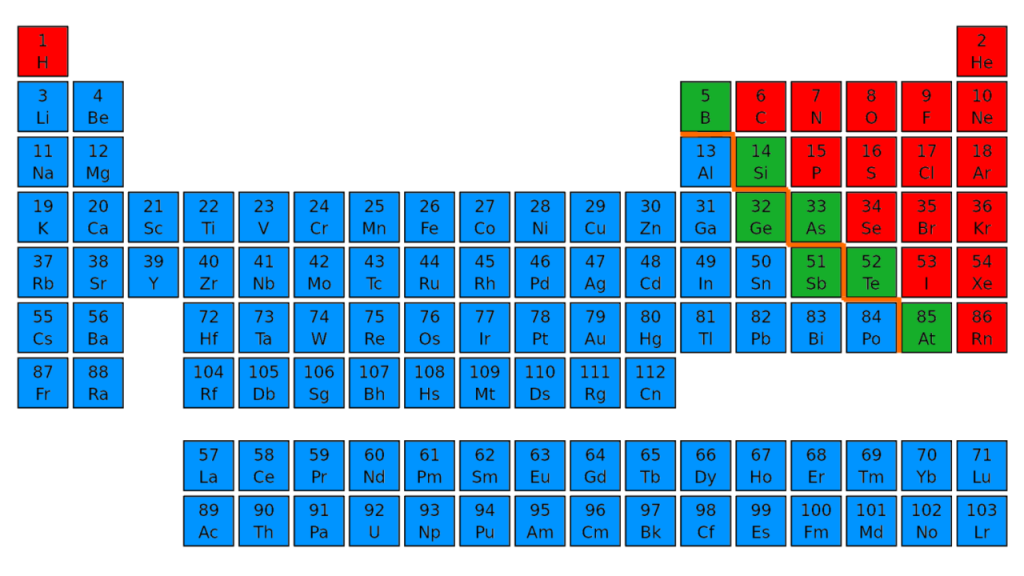
Conduction: Some metalloids, such as silicon and germanium, can act as electrical conductors under the right conditions, thus they are called semi-conductors. Physical Properties of Metalloids State: They are all solid at room temperature. Silicon – a shiny grey solid, an electrical conductor and has a high melting point (metallic properties) but is very brittle and has a low density (non-metallic properties). Metalloids are useful in the semiconductor industry.Carbon – a dull, dark grey solid and is brittle (non-metallic properties) but has a high melting point and is a conductor of electricity and heat (metallic properties).These elements are found close to the zig-zag line that separates the metals from the non-metals. These elements are called metalloids or semi-metals. These individual element summary pages contain a lot of additional information as well as references. When exploring the table or list views on this page, please note the links to dedicated pages for each element. Observation, classification and analysis skills will be mature. PubChem is providing this periodic table page in order to help navigate abundant chemical element data available in PubChem. 
To note trends for the metallic eigenschaft of elements. To discover and arrangement of minerals, non-metals and metalloids switch the periodic table. Some elements between the metals and non-metals in the periodic table have properties which are a mixture of the properties of metals and non-metals. To become familiar with the properties of metals, non-metals and metalloids. When non-metals are in a solid state, they are usually brittle so you can’t beat them into shape. Many non-metals have low melting and boiling points. Graphite (a form of carbon) is a rare example of a non-metal that conducts electricity very well. Non-metals have a variety of properties, but very few are good conductors of electricity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
